Montana IPM Bulletin, Fall 2016

The Montana IPM Bulletin presents critical pest management and pesticide education articles for Montana homeowners, pesticide applicators, farmers and ranchers. These articles are designed to deliver timely updates from an unbiased perspective that are specific to Montana. This is a cooperative effort between Montana State University pesticide education and integrated pest management programs.
In this Issue
- Fall is a Great Time to Control Pocket Gophers
- Noxious Weeds and Montana Grazing Lands: An Economic Perspective
- Management of Glyphosate (RoundUp and generic products) Resistant Kochia with Soil Applied Herbicides
- Fumigants offer Unique Approach to Managing Rodents and Structural Insects
- Management of Wheat Streak Mosaic Virus in Cereals Requires Integrated Pest Management
- Ask the Expert
- Meet Your Specialist
- Pest Management Toolkit
- Acknowledgments
PDF links can be viewed with Adobe Acrobat or Sumatra PDF.
Fall is a great time to control Pocket Gophers
Stephen Vantassel, Vertebrate Pest Specialist, Montana Department of Agriculture

Montana is home to only one pocket gopher species, the Northern Pocket Gopher (Tomomys talpoides). Like other species of pocket gophers, Northern Pocket Gophers are vegetarians, feeding on tap-rooted plants, such as forbs, tree roots, and alfalfa.
Pocket gophers can significantly hamper agricultural production in three ways. First, pocket gophers feed on the roots and stems of plants. They also clip plants and store them below ground for later consumption. Second, pocket gophers bury plants using the soil excavated during their burrowing activities. In fact, one study found that a single pocket gopher can move 254 cubic feet of soil, per acre, per year. The surface mounds can increase bare ground from 5-25 percent depending on density and activity of the pocket gophers present (Figure 1). The bare soil also allows invasive plants to obtain a foothold in the field. Finally, pocket gopher mounds require harvesters to cut plants above the level of the mounds or suffer dulled cutting surfaces or equipment breakage.
So, how bad can pocket gopher damage be? Studies have found that pocket gophers can reduce rangeland production between 18-25 percent. In poundage terms, researchers in California determined that pocket gophers can reduce rangeland production by 250 pounds per acre per year.
While all this information may be depressing for those with pocket gophers, be assured that all is not lost. Researchers also found that control of pocket gophers can improve forage production by 16 percent within two months of control.
Assessment
When looking over a field covered in pocket gopher mounds, it is easy to become overwhelmed and think that there are too many pocket gophers to control. Research shows that pocket gopher numbers can range from 19 per acre in poor habitats like grasslands to 73 per acre in great habitats like alfalfa fields. But most producers will have 24- 36 per acre in areas with long-standing pocket gopher activity. We encourage producers to keep pocket gopher numbers at low levels to improve production. Alfalfa growers should be particularly vigilant in their effort to manage pocket gopher populations.
While the number of mounds make it appear that pocket gophers occur throughout the field, closer examination will reveal that they are only active in certain areas. Active pocket gopher areas are indicated by fresh mounds. Fresh mounds differ from old (inactive areas) by their darker color (indicating higher moisture content), height, and finer granularity of the soil (it isn’t as clumped as older mounds). If it is difficult to identify the active areas, use a harrower to knock down all the mounds so that new mounds will be located easily.

Figure 1. Pocket gopher damage. Photo by Stephen Vantassel.
Timing
While pocket gophers may be controlled whenever fresh mounds appear, control efforts should be planned for the two main peaks of pocket gopher activity, namely spring (when the ground thaws) and fall (before the ground freezes).
Spring control removes pocket gophers before reproduction, thereby eliminating the female and her offspring. Fall control removes yearling pocket gophers that have recently established territories, which may include your field.
Control Methods
Producers frequently want to know what the “best” method is to control pocket gophers. Unfortunately, the answer depends on the situation and goals. I suggest that producers think in terms of economic benefit. To achieve total eradication of pocket gophers in a field, the effort may cost more than the entire revenue of the field. think less about the “best” method and think instead of the “most cost-effective” method. The following information is provided to help make a cost-effective decision.
General Principles
As a rule, toxic baits, such as those made with zinc phosphide, strychnine, chlorophacinone and diphacinone, are the most cost- effective way to control pocket gophers. Toxic baits are relatively inexpensive and take relatively little time to apply. For example, two people needed only two hours to hand-bait about five acres for pocket gophers. We were not working very quickly and even took the time to knock down the mounds after application.
Try to use a different active ingredient every several years or so to reduce the chances of bait shyness and resistance. Don’t ignore the benefits of trapping. While trapping is not cost-effective as the first line of control for large acreages (15 or more), it can be quite helpful as a follow up to toxicants. Plus it’s a great activity for teenagers to learn.
The trick to effective control of pocket gophers is consistency and persistence. Two to three years of treatment should provide signifcant results allowing you to then move into reassessment mode that may not include continued treatment. Alfalfa growers are an exception. They should expect to apply significantly more effort to achieve desired control. For more information on rodent management see the Montana Department of Agriculture website.
Noxious Weeds and Montana Grazing Lands: An Economic Perspective
Kate Fuller, Extension Economics Specialist; Stacy Davis, Research Associate; Jane Mangold, Extension Extension Invasive Plant Specialist
Successful management of invasive plants is critical to maintaining healthy agricultural and wildland systems and relies on adequate funding. Adequate funding to achieve such goals, however, hinges on a clear understanding of what is being lost to weed invasions – for example, livestock forage production – and what is being spent to control weeds.
Published information on the economic losses caused by invasive weeds is scarce. The most recent, in- depth economic analyses of noxious weeds in Montana occurred some 20 years ago. In one analysis, the direct economic costs of spotted, diffuse, and Russian knapweed on range and wild lands amounted to over $14 million annually [expired link]. In another analysis, the direct and secondary economic impacts of leafy spurge in Montana totaled about $19 million [expired link].
We revisited the topic of the studies mentioned above by developing a 16-question survey concerning noxious weed management and associated costs. The target audience for the survey was livestock producers who were grazing livestock on privately-owned rangeland in Montana. Survey responses were received from 129 people in 45 counties in Montana, with the majority of respondents grazing cattle (88%) followed by sheep and horses (29% and 23%, respectively). The majority of the survey referred to the largest contiguous block of privately owned or leased land on which respondents graze livestock, which we refer to as “Block A.” The average size of Block A was 5,055 acres.
The most common noxious weeds were Canada thistle, leafy spurge, and houndstongue. However, leafy spurge, Canada thistle, and knapweed (spotted and diffuse) were reported as causing the largest decreases in livestock production. Only 6% reported having no noxious weeds on Block A. Using information from other studies where field data estimated forage loss due to two state-listed noxious weeds, we estimate the average reduction in biomass resulting from the reported presence of spotted knapweed and leafy spurge at 0.7 and 0.8%, respectively. We estimate the corresponding average value of the reduction in stocking rate is $0.40 per acre per year, or $2,022 for the average Block A.
Respondents listed the most commonly used strategies to prevent noxious weeds from establishing as monitoring and grazing management (grazing to maintain competitive vegetation). Only 3% indicated using no management strategies to prevent noxious weeds from establishing. The top three strategies used to control established noxious weeds on Block A were chemical control (88% of respondents), grazing (29% of respondents), and biological control (27% of respondents). About 46% of respondents utilized more than one control strategy or integrated weed management.
Respondents’ average total cost of noxious weed control, including labor and materials, was estimated to be $0.89 per acre per year, or $4,499 per year for the average Block A. However, costs of noxious weed control ranged a great deal across individuals – from $0 to over $40 per acre. We estimate the total economic loss over all of Block A land, including both the costs of control and the costs of foregone production, to be $1.29 per acre per year, or $6,521 per year for the average Block A. However, it is important to note that Block A acreage represents only a small percentage of privately owned land in Montana. In addition, the per-acre numbers would undoubtedly be higher if we were to incorporate reduction in biomass production resulting from weeds other than leafy spurge and spotted knapweed, the two species for which we have biomass reduction models.
In addition to questions regarding economic losses, respondents were asked to indicate their level of agreement with a series of statements regarding Block A. Approximately 63% of respondents either agreed or strongly agreed with the statement that noxious weeds are a problem for them despite their efforts to manage them. The majority of respondents (68%) agreed that noxious weeds are a problem for them because they are spreading from their neighbor’s land to their own land; a smaller majority (57%) agreed that noxious weeds are a problem for them because of their potential to spread onto their neighbor’s land and cause conflict. Only 16% of respondents felt that noxious weeds are only a problem for them because of regulations that say they must control noxious weeds and not because they cause a reduction in grazing capacity.
The project was funded with a grant from the Montana Noxious Weed Trust Fund (MDA Grant 2015-006).

Management of Glyphosate (RoundUp and generic products) Resistant Kochia with Soil Applied Herbicides
Ed Davis, Research Associate; Tim Seipel, Research Scientist; Fabian Menalled, Crop Weed Specialist
Kochia (Kochia scoparia (L.) Schard.) is an early germinating summer annual broadleaf weed commonly found in Montana’s crops, pastures, and roadsides. It is a self- and cross- pollinated species with pollen moving between plants on the wind. Each kochia plant can produce over 10,000 seeds. Kochia has a unique seed dispersal mechanism; after a plant has matured, as a tumble-weed, it breaks of at the ground and rolls in the direction of a slope or wind. These traits could result in a rapid spread of plants. Another problem that further complicates kochia management is the selection of herbicide-resistant biotypes.
|
Site Location |
Soil Texture |
Organic Matter (%) |
pH |
|---|---|---|---|
|
Bozeman |
Clay Loam |
2.3 |
7.1 |
|
Denton |
Sandy Clay Loam |
3.2 |
7.7 |
|
Gilford |
Loam |
4.2 |
7.8 |
|
Havre |
Loam |
1.8 |
7.9 |
|
Miles City |
Loam |
2.2 |
8.2 |
|
Townsend |
Loam |
2.1 |
7.9 |
Table 1. Site locations and soil characteristics at each site where herbicide trials were conducted.
|
Herbicide |
Herbicide Group |
Rate of Application (oz/acre) |
|---|---|---|
|
Fall Applied |
||
|
Metribuzin + Spartan |
5 & 14 |
8 + 4 |
|
Spartan Charge |
14 |
7.62 |
|
Valor |
14 |
4 |
|
Spring applied |
||
|
Authority MTZ |
5 & 14 |
12 |
|
Spartan Charge + Metribuzin |
14 & 5 |
6.34+8 |
|
Spartan Charge |
14 |
6.34 |
|
Valor |
14 |
3 |
Table 2. List of soil applied herbicides with potential for controlling kochia in either fall or spring. At the six tested sites, all these products or product combinations controlled over 90% of kochia.
Herbicide resistance results from repeated use of the same herbicide and is the innate ability of a weed biotype to survive and reproduce after treatment with an herbicide dose that would normally be lethal. In Montana, the first herbicide-resistant biotypes of kochia were found in 1984 when individuals were not controlled with Group 5 Photosystem II inhibitor herbicides such as atrazine. In croplands, kochia resistance to Group 2 ALS inhibitors herbicides including chlorsulfuron (Glean, Telar) and metsulfuron-methyl (Escort) was detected in 1989. In 1995, kochia resistance to Group 4 Synthetic Auxins herbicides including dicamba (Banvel) and furoxypyr (Starane) was confirmed. The first kochia biotypes resistant to Group 9 EPSP synthase inhibitors such as glyphosate (RoundUp and other generic products) were confirmed in Montana in 2012. More recently, multiple resistance in kochia to both Group 2 ALS inhibitors herbicides and glyphosate were found.
Herbicide resistance results from repeated use of the same herbicide and is the innate ability of a weed biotype to survive and reproduce after treatment with an herbicide dose that would normally be lethal. In Montana, the first herbicide-resistant biotypes of kochia were found in 1984 when individuals were not controlled with Group 5 Photosystem II inhibitor herbicides such as atrazine. In croplands, kochia resistance to Group 2 ALS inhibitors herbicides including chlorsulfuron (Glean, Telar) and metsulfuron-methyl (Escort) was detected in 1989. In 1995, kochia resistance to Group 4 Synthetic Auxins herbicides including dicamba (Banvel) and furoxypyr (Starane) was confirmed. The first kochia biotypes resistant to Group 9 EPSP synthase inhibitors such as glyphosate (RoundUp and other generic products) were confirmed in Montana in 2012. More recently, multiple resistance in kochia to both Group 2 ALS inhibitors herbicides and glyphosate were found.
Controlling glyphosate resistant kochia in non-crop fallow periods with soil residual herbicides provides an opportunity to rotate herbicide site of actions, reducing the pressure towards the selection of herbicide-resistant biotypes. To ensure that soil active herbicides are in place prior to kochia emergence, they have to be applied in the fall as a post-harvest treatment, or early spring, prior to kochia emergence. The goal of these applications is to keep the fallow area free of kochia from April through mid- July as its germination and establishment is unlikely after mid-July.
Between 2013 and 2015 with support from the Montana Wheat and Barley Committee, we assessed the potential of soil-applied herbicides to control glyphosate resistant kochia. To encompass a wide range of environmental conditions, we selected six sites across Montana (Table 1) and tested 17 different herbicide combinations that were applied in late fall 2013 or early spring 2014. We then seeded either spring wheat or winter wheat to assess crop safety.

Although we observed site to site differences in herbicide performance, of the 17 different herbicides screened, three herbicides or herbicide mixtures (Metribuzin + Spartan, Spartan Charge, and Valor) controlled over 90% of kochia (Table 2) in fall applied post- harvest applications. Four herbicide or herbicide mixtures (Authority MTZ, Spartan Charge + Metribuzin, Spartan Charge, and Valor) controlled over 90% of kochia in spring applications, while causing no damage and not reducing yield from residual effects (Table 2). The different herbicide mixtures had different herbicide groups (modes of action) when compared to those typically used during the crop phase. These herbicides give farmers more options in controlling kochia through the fallow phase and increase the diversity of herbicides currently used.
Reducing the risk of selecting for herbicide-resistant biotypes requires producers to routinely scout prior to and following each herbicide application. Producers should also rotate management practices. For example, crop rotation is an excellent tool to reduce the selective pressure on herbicide resistant weeds.
More information on herbicide resistance can be found in the MSU Extension MontGuide, Preventing and Managing Herbicide-resistant Weeds in Montana (460KB PDF).
Fumigants offer Unique Approach to Managing Rodents and Structural Insects
Cecil Tharp, MSU Extension Pesticide Education Specialist
Broadcast sprays, baits and seed treatments are quick and effective go-to pesticide strategies; however fumigants are another excellent tool often overlooked for managing rodent and insect pests. In Montana, phosphine gas (PG), chloropicrin and methyl bromide are all fumigants that can be used to fumigate empty structures; however the federal government has banned the importation and production of methyl bromide since January 1, 2005, due to its depletion of the ozone layer. This means much higher prices with the use of methyl bromide. In addition, chloropicrin cannot be used in storage buildings and bins which contain an agricultural commodity due to persistence on stored grains. Therefore the PG fumigants, aluminum phosphide and magnesium phosphide, are the most common fumigants for managing insect and rodent pests in Montana. Aluminum phosphide is a grayish-green solid which when exposed to moisture, will react to produce hydrogen phosphide, more commonly known as phosphine gas.
Examples of fumigant products with magnesium or aluminum phosphide:
- Degesch Fumi-cel
- Fumitoxin
- Degesch Magtoxin Granules
- Degesch Fumi-Strip
- Detia Fumex
- Phosfume 2
- Degesch Phostoxin
- Weevil-cide
Special training is needed to use PG fumigants as these fumigants are federal restricted-use pesticides due to high acute toxicity when inhaled. It is critical that applicators read and understand the pesticide product label before applying these products.
Structural Insect Pests
The Grain Inspection Packers and Stockyards Administration (GIPSA) and the Food and Drug Administration (FDA) have standards to determine if an infestation is occurring. If grain is identified as infested, grain elevators often won’t accept delivery of the grain or it will be labeled as “sample grade.” Sample grade grains are often given a much lower price.
Some thresholds:
- If two or more live insects (that are injurious towards grain) are found per kilogram (2.2 lb) seed, the grain is infested according to GIPSA standards and labeled as “sample grade.”
- If there are greater than 31 insect damaged kernels of wheat per 100 grams (0.22 lbs), the wheat is labeled as “sample grade.”
Phosphine gas fumigants can effectively kill insects that are contaminating grain. These fumigants evenly distribute within a grain bin due to the pesticide’s high vapor pressure. This ensures high efficacy even if the bin is filled with an agricultural commodity. Before using PG fumigants, applicators should identify pests that are harmful to the agricultural commodity stored in the structure. Montana applicators can use the Pest Management for Grain Storage and Fumigation (3.2MB PDF) for assistance identifying grain insects. Complete kill of insects is possible, but phosphine gas needs an extended period of time to be effective (depending on temperature and humidity).
Always read and follow all product label requirements, however some important reminders include:
- For stationary structures, phosphine readings must be conducted from within structure in order to characterize the application, determine the fumigator’s exposure and necessary personal protective equipment.
- All phosphine concentration readings must be documented.
- If monitoring equipment is not available, an approved canister respirator must be worn during application within an enclosed area.
- All entrances to the fumigated structure must be placarded until phosphine readings are below safety thresholds outlined on the pesticide product label.
Rodents
Fumigants such as ignitable gas cartridges and PG fumigants can be effective for managing burrowing rodents, however they are usually a follow-up to poison baits (see article page 1). Phosphine gas products are sometimes preferred due to the risk of fire and personal injury using ignitable gas cartridges. Applicators can increase efficacy of PG fumigants by 1) crumpling newspapers and inserting into active burrows, 2) covering burrow openings and packing with soil, 3) inserting a length of plastic pipe (1 1/2’” x 1 ¾” diameter) through soil into burrow, and finally 4) placing 2 to 4 tablets or 10 to 20 pellets in each active burrow through the plastic pipe. Applicators should also be aware of a few additional product label requirements active since 2012.
Additional product label requirements include:
- Rodent fumigations must occur only on burrowing pests in agricultural areas, orchards, non-crop areas (such as pasture and rangeland), golf courses, athletic fields, parks and recreational areas, cemeteries, airports, rights-of-way, earthen dams, and other non-residential institutional or industrial sites.
- For burrowing pest applications, products must not be applied in a burrow system that is within 100 feet of a building that is or may be occupied by people or domestic animals. This buffer zone for treatment around non-residential buildings that could be occupied by people or animals has been increased from 15 feet to 100 feet.
- Fumigation Management Plans must be written before all applications of phosphine products, including all burrowing pest fumigations.

Photo courtesy of Degesch America
Always read and follow the product label requirements. For more information on managing rodents see the Montana Department of Agriculture pest bulletins. For more information on managing rodents contact Stephen M. Vantassel, Vertebrate Pest Specialist, (406) 538-3004, [email protected]
Fumigant Training across Montana
Phosphine fumigants are an effective management tool; however if used improperly they can be extremely dangerous. There have been at least five deaths in the United States and Canada since 2000 from inhaling phosphine gas. These incidents have spurred EPA and product registrants to implement new restrictions regarding phosphine fumigants since 2012.
The Montana State University Pesticide Education Program (MSU PEP) is offering three fumigant training across Montana: February 21 (Malta), February 22 (Lewistown) and February 23, 2017 (Choteau). Private and commercial applicator credits will be offered for attending either the morning session focused on rodent fumigations or the afternoon session focused on structural fumigations. Applicators can view the complete agenda by navigating to the MSU PEP website and selecting the ‘Fumigant Training Tour’ from the rotating training carousel.
Further Information
If you have questions regarding this article or aluminum and magnesium phosphide, contact Dr. Cecil Tharp (MSU Extension Pesticide Education Specialist, (406) 994-5067, [email protected]). For more information regarding the fumigant training opportunities or to pre-register, contact Amy Bowser (MSU Pesticide Education Technician, (406) 994-5178, [email protected]).
Management of Wheat Streak Mosaic Virus in Cereals Requires Integrated Pest Management
Mary Burrows, Extension Plant Pathologist and Zach Miller, Assistant Professor and Western Ag Research Center Superintendent
Wheat streak mosaic virus (WSMV) in cereals was a major issue in Montana in 2016, where it was identified in 24 counties. Annual losses due to WSMV are typically minor, averaging around 1% of wheat yields across the Great Plains, but were much higher in Montana during 2016. A combination of factors favored a WSMV epidemic including abundant volunteer wheat due to widespread hail in 2015 and reduced control of volunteer wheat, early planting dates to achieve high yields, timely rains which allowed early planted wheat to germinate and grow, and a long extended fall and open winter which favored reproduction and spread of the wheat curl mite (WCM) that vectors WSMV. In 2016 we observed widespread destruction of infected winter and spring wheat crops and yield losses in severely affected areas. Even organic production, which is normally protected from this disease due to intensive tillage and crop rotation, has been impacted by this disease due to the widespread nature of the problem.
Montana State University has been working on WSMV and other WCM-transmitted viruses since 2007, along with the University of Nebraska, Kansas State University, Texas A&M, and other cooperators. We are currently involved in a cooperative effort to develop models that will predict these epidemics with better accuracy, but there are several known risk factors and management practices that can be integrated to reduced risk of WSMV in cereal crops. WSMV and the WCM can only survive on living grasses and thus need a ‘green bridge,’ to be perpetuated from harvest to the new crop (Figure 2). Traditionally, the green bridge is made up of wheat seed knocked to the ground by hail that then germinates. This is also called a ‘volunteer’ crop. Grassy weeds such as downy brome that host the vector and disease can also create a green bridge between crops. The wheat stem sawfly (WSS) is an often underappreciated, but serious contributor to the green bridge. The insect cuts stems at crop maturity, leading to wheat stems falling to the ground, with resulting seed shatter and subsequent germination (volunteer).
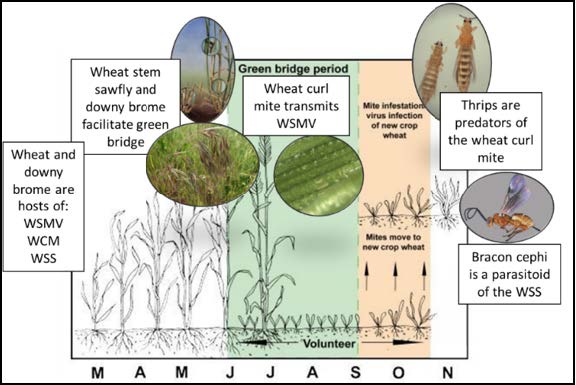
Figure 2. The green bridge occurs when green plant material is present between planting and harvest. This is facilitated by wheat stem sawfly and downy brome (cheatgrass). Thrips and parasitoids are biocontrols for wheat curl mites and wheat stem sawfly.
The most reliable management option for WSMV and the WCM is eliminating the green bridge. This is accomplished both by 1.) terminating any green material in the field and surrounding area with herbicide or tillage at least 2-3 weeks prior to planting winter wheat, and 2.) avoiding planting too early in the fall. Most mites are moving during the harvest period as they leave the maturing crop. Colder temperatures reduce mite reproduction, movement and spread from source plants. A hard frost can significantly reduce mite numbers (Figure 3), but in the absence of a frost event, the mites continue to grow and reproduce on alternative (non-crop) host plants. Fall weather also affects WCM survival as they are moving between plants. At high temperatures (30˚C, 86˚F) and low relative humidity, mites survive off a host plant only a few hours. At low temperatures (10˚C, 50˚F) and high relative humidity, mites survive off a host plant for 5-7 days. Relative humidity seems to be more crucial than temperature. Risk may also increase in areas where winter and spring cereal crops overlap in the same area, and when a hail event causes widespread cereal crop volunteer. While delaying planting of winter wheat can help reduce the risk of plants getting infected, the opposite tactic works in the spring. Early planting of spring wheat, durum and barley reduces the risk of plants getting infected due to colder temperatures and increased plant age when the mite movement starts from winter wheat or alternative hosts.
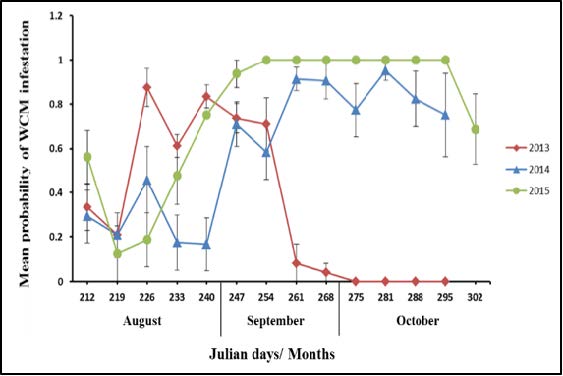
Figure 3. Wheat curl mites in Bozeman are reduced by frost events (2013) but otherwise continue landing on spring wheat well into the fall (2014 and 2015).
Host resistance is an excellent way to manage WSMV and the WCM. Unfortunately, there are no varieties resistant to either the virus or mite currently available in Montana. There seems to be some tolerance in winter wheat varieties, but spring wheat varieties are highly susceptible. We have little data on barley, but varieties we have tested are susceptible. Yield loss is not directly related to symptom severity, but can be related to the timing of infection. Early infections, including fall infection, have much greater effects on yield than infections later in the season. Wheat infected after heading will likely experience very little loss due to WSMV. WSMV reduces root growth, which then leads to early drought and, very commonly, blank heads or light grain. This also stresses the plant and causes mites to leave the host plant.
There are no insecticides labeled for WCM management. At Montana State University, we evaluated multiple pesticides for control of WCM including organophosphates, carbamates, pyrethroids, mite growth inhibitors, soaps, oils, contact pesticides, and plant defense initiators. One active ingredient provided some control of the WCM but did not reduce spread of WSMV. This product is not labeled for WCM. Insecticides also kill non-target insects including thrips, which are predators of the WCM. This may increase disease risk. Mites die without a living host plant to feed on. In order of efficacy for host plant termination and reduction of mite survival, tillage >> paraquat > glyphosate >>> grazing.
Grazing is often suggested as an option for green bridge management, but plant termination does not occur fast enough, cow lips are not close enough to the ground to graze all plant material out of the field, and not all plants are terminated. Epidemics are often associated with incomplete plant kill and extension of the green bridge due to grazing. Herbicides can be used on a field prior to grazing, but check labels for grazing restrictions.
Ask the Expert
Q: I’ve heard that there has been quite a bit of white mold this year in northeastern Montana. Why has this year been so bad?
Jessica Rupp says: It has been a difficult year for white mold in the pulse crops. All the elements required to have disease were present, including the susceptible crops of pea, chickpea, and lentil, the Sclerotinia white mold, and an incredibly conducive environment. Greater than average rainfall resulted in an environment that was perfect for fungal growth. This became quite a problem in lentils in northeast Montana. White mold infection usually begins in dying petals and moves into the plant. The disease is most common when the canopy has reached closure and limited airflow provides a humid environment for fungal growth. Fluffy, white mycelial growth occurs at the base of the plant, on stems, and even pods. The fungus produces sclerotia, which resembles mouse droppings. Sclerotia are capable of surviving in soil in harsh conditions for many years. Once white mold gets started it is hard to control due to the difficulty of fungicides reaching the affected areas. Sclerotinia is able to survive in soil for several years, so a rotation to non-host crops, such as cereals, is recommended for at least four years. With the rapid growth of pulse acres in Montana, it is especially critical to prevent inoculum build-up. In certain high-risk areas, producers next year might consider a protective fungicide. Seed testing is also important to ensure clean seed is being planted. Also, any fields near last year’s should be monitored closely.
Q: I am thinking about purchasing a Pressurized Exhaust Rodent Control (P.E.R.C.) system to control my pocket gophers. Is it an effective way to control pocket gophers?
Stephen Vantassel says: The device has not been tested in Montana. P.E.R.C. system, manufactured by H & M Gopher Control of California, uses a gasoline engine to produce carbon-monoxide gas which is then pressurized for injection into rodent burrows. A study by the University of California used the device to control pocket gophers in alfalfa fields. Using a 2 minute injection time, researchers achieved control of 61%, 63% and 45% in the three tests. Researchers speculated that the low efficacy achieved in the third test was due to operator inexperience and poor soil conditions which prevented the gas from reaching toxic levels. The device can be quite expensive depending on the features. However, it is a fast way to treat pocket gopher burrows, taking less time than trapping and aluminum phosphide. Carbon-monoxide is quite toxic. So, use carefully and avoid applications within 100 feet of structures.
Q: What grasses do you recommend seeding after I control my weeds (cheatgrass and spotted knapweed)? I live in the southern portion of the Bitterroot Valley.

Figure 4. Bluebunch wheatgrass plants that were seeded into spotted knapweed infestation. Photo by Jane Mangold.
Jane Mangold says: It’s hard to give a one-size-fits all prescription for what to seed after weed control. Species selection depends first and foremost on land management goals (forage/livestock production, wildlife habitat, aesthetic, slope stabilization, etc.). Then, it depends on things like precipitation, soil texture, aspect, elevation, etc. In general, non-native grasses tend to do much better than native grasses, but again the choice depends on management goals and degree of degradation at the site.
Based on some of my revegetation research in western Montana, I like tall wheatgrass (non-native), bluebunch wheatgrass (native) (Fig. 4), intermediate and/or pubescent wheatgrass (non-native), and Basin wildrye (native, fairly specific habitat). Other people tell me green needlegrass (native), Russian wildrye (non-native), and sheep fescue (non- native) are good candidates. A couple useful Extension publications to help you with this question include Revegetation Guidelines for Western Montana (PDF) and Dryland Pastures in Montana and Wyoming (1.4MB PDF). In addition to your local Extension office, another great resource is the Natural Resources Conservation Service (NRCS).
Q: Can I control Canada thistle in my garden using acetic acid (vinegar)?
Fabian Menalled says: Canada thistle is a rhizomatous, perennial, herbaceous plant that has been classified in Montana as a Priority 2B noxious weed. It grows 2 to 5 feet tall but can develop an extensive root system, which can spread 10-12 feet or more in just one season. Acetic acid burns (desiccates) top growth and because it does not translocate, it does not kill underground roots. Usually, Canada thistle quickly replaces the burnt tissue with new top growth. Theoretically, one can continue to burn off top growth and starve the roots, but that would take years and require fastidious attention to timely and repeated applications. Canada thistle is difficult to control once it is established on a site. Thus, prevention and early detection are essential. One treatment or a combination of treatments may need to be used to control populations depending on the site. You can find more info about the ecology and management of Canada thistle in the USDA Invasive Species Technical Note, Ecology and Management of Canada thistle (1MB PDF).
Q. Am I at risk of poisoning from my county’s mosquito fogging program?
Cecil Tharp says: If used properly, no. Counties are generally responsible for managing mosquito populations for residents. This can be a difficult task in areas of the state with standing water where mosquitoes thrive. The most common choices for managing mosquitoes are early season larvicides and late season fogging (otherwise known as a type of ultra-low-volume application (ULV)). Larvicides target the immature stages of mosquitoes early in the year using insect growth regulators or bacterial insecticides that have very little toxicity towards mammals. ULV applications later in the year target adult mosquitoes using malathion, naled or pyrethroids.
According to EPA, each ULV product can be used for public health mosquito control programs without posing unreasonable risks to the general population when applied according to the label. There are reports of individuals with chemical sensitivities exhibiting minor symptoms including runny nose, itchy eyes and throat, consequently they should lower exposure by going inside while applications are occurring. It is possible for an individual to experience more severe symptomology if pesticide products are applied well above the labeled product rate and individuals are directly exposed. This is quite rare. County or commercial pesticide applicators must hold a commercial/ government applicator license that ensures they are trained to apply pesticides at the proper timing and rates.
If you would like more information or suspect pesticide poisoning contact Cecil Tharp (406-994-5067 or email at [email protected]).
Meet Your Specialist
Amy Bowser, Pesticide Education Technician, Pesticide Education Program

Where/when did you receive your degrees?
I graduated from Montana State University in 2009 with a Bachelor’s degree in Environmental Science with an emphasis in soil and water. After graduating I moved to Anchorage, Alaska, and obtained a Master’s degree in Environmental Science from Alaska Pacific University in 2012 where I focused on glacial ecology.
What is your field of interest?
I enjoy studying natural sciences including ecology and environmental systems. My academic background has ranged from research on mulches to studying glacial algae. Currently, in my position with the Pesticide Education Program, I am focused on learning about pesticides in the environment.
When did you arrive in Bozeman?
I moved to Bozeman as a freshman in college in 2005. I left in 2010 for graduate school, but returned in 2012. I moved to Livingston, in 2014.
Where are you from originally?
I was born and raised in Eagle River, Alaska, but moved around with a few years in Oregon and Washington. My family ultimately ended back up in Alaska.
Where have you worked/taught in the past?
Prior to my position with the Pesticide Education Program, I worked for several years at an environmental engineering and management firm in Bozeman. I was responsible for analyzing water quality data from several Superfund sites to assist remediation efforts and supported the environmental group with environmental site assessments, wetland delineations and site characterization. I gained a wide range of knowledge of how different materials from metals to petroleum products move and influence the environment. This is directly applicable to understanding pesticide movement and impacts within the environment.
What are your hobbies?
For the past few years I have been spending most of my weekends remodeling my house! When I can get away, my summers are spent experimenting with native plants in my garden, floating the Yellowstone River and exploring Montana’s wilderness areas and national forests. Winters involve skate skiing, hanging out at Forest Service cabins and visiting as many hot springs as possible.
What does your position entail?
I started in May 2016 as the Pesticide Education Technician with the Pesticide Education Program. Since May I have been learning about everything to do with pesticides! My tasks have ranged from support of the Private Applicator Program to updating MontGuides and our website. I am also starting to put together presentations for multiple educational events coming up this winter.
Pest Management Toolkit
The Montana Department of Agriculture has revised several of its Vertebrate Pest Control Bulletins
Revised Bulletins include, Prairie Dogs, Pocket Gophers, Registered Rodenticides for Field Rodents, Richardson’s Ground Squirrel, and Prevention of Woodpecker Damage. These bulletins are available in PDF format for immediate download, find them on the Montana Department of Agriculture Vertebrate Pests page.
New Vertebrate Pest Specialist
The Montana Department of Agriculture has hired Stephen M. Vantassel as its Vertebrate Pest Specialist. Stephen comes from the University of Nebraska-Lincoln where he spent 10 years as an Extension Program Coordinator handling wildlife damage related issues.
Stephen is available to provide consultation, training, and additional support for Montanans dealing with vertebrate pests. He can be reached at (406) 538- 3004, [email protected] or by visiting his office at 625 NE Main St., Ste 3, in Lewistown, Montana.
Two new weed-related publications from Montana State University Extension
- Biology, Ecology, and Management of Curlyleaf Pondweed (2.5MB PDF) provides information on a Priority 2B noxious aquatic weed. Curlyleaf Pondweed is an aquatic invasive plant first reported in Montana in Lake County in 1973. Learning to identify curlyleaf pondweed and reporting it when found as well as preventing unintentional transfer from infested to non-infested waters is paramount. Printed in color with photos, 10 pages.
- How to Prevent Non-target Injury of Broadleaf Crops and Vegetables by Residual Herbicides (2.4MB PDF) is a fact sheet to help homeowners and herbicide applicators avoid issues with persistent herbicides stemming from contaminated soil amendments. Color, 2 pages.
Northern Rockies Invasive Plant Council conference
- October 17- 20, 2016, at the Boise Centre, Boise
- Symposia on Russian olive, invasive mustards, and rush skeletonweed; keynote presentations from Roger Sheley, Jeanne Chambers, Daniel Tekiela, Matt Germino, Urs Schaffner, and John Proctor; and breakout sessions.
- Visit nripc.org for more information.
Montana Weed Control Association annual conference
- January 10-12, 2017, at the Heritage Inn in Great Falls.
- Visit mtweed.org for more information.
Montana Noxious Weed Identification App
- Visit EDDMapS West to read more and access the app through the App Store or Google play.
National Weed Survey
The National and Regional Weed Science Societies conducted in 2015 a survey of the most common and troublesome weeds in 26 different cropping systems and natural areas across the U.S. and Canada. The survey compiles were nearly 700 responses from weed science society members from 49 states, Puerto Rico, and eight Canadian provinces. More than 650 weed species were mentioned at least once by survey respondents, and Montana’s weeds such as kochia, Canada thistle, and foxtails are listed among the top 10 most troublesome and common weeds.
The entire data set is available for download (87KB Excel Spreadsheet)
Newly Designed and Updated Cropland Weed Ecology and Management Extension Website
- Visit the new site at ipm.montana.edu/cropweeds
The Weed Science Society of America published Facts about Weeds
- A brochure that includes dozens of interesting and informative tidbits – from weed seeds found near outer space to the impact of weeds on crop production. The PDF version is available for download (260KB).
2017 Pesticide Safety Education Update
- Bozeman, MT. April 21st - 22nd
- This event is open to pesticide trainers across Montana. The event covers new technologies, new certifications and training requirements, and novel approaches to teaching pesticide core areas.
- Pre-register by contacting Amy Bowser ([email protected]; 406-994- 5178). Pre-registration fee is $150. You can send payments written to the MSU Pesticide Education Program; send to Attn: Amy Bowser, 103 Animal Bioscience, Montana State University, Bozeman, MT 59717-2900.
Fumigant Training Tour
- Malta, Lewistown, and Choteau. February 21-23, 2017
- These programs will focus on the proper use of fumigants when managing rodents and structural insects. The Malta program is on February 21, Lewistown program is on February 22 and the Choteau program is on February 23. Commercial and private applicator credits will be offered. Pre-registration is mandatory.
- View the agenda by navigating to pesticides.montana.edu and select fumigant training tour from the rotating carousel. For more information contact Amy Bowser at (406) 994-5178.
Rodent Pest Control Training
- Bozeman, MT. October 17
- Individuals interested in learning about managing rodent populations should attend this event. This program will offer commercial and private applicator credits, while bringing in the MDA Vertebrate Pest Specialist to present various rodent management techniques.
- For more information or to pre-register contact Theresa Schrum (MT Dept. of Agriculture (406) 587-9067 or [email protected]) or see the complete agenda on the Private Applicator Program webpage, select “Training Opportunities” then “Region 2.”
Acknowledgments
Do You Have a Question or Comment Regarding the Montana IPM Bulletin?
Send inquiries and suggestions to:
Cecil Tharp
Pesticide Education Specialist
P.O. Box 172900
Montana State University
Bozeman, MT 59717-00
Phone: (406) 994-5067
Fax: (406) 994-5589
Email: [email protected]
Web: pesticides.montana.edu
Jane Mangold
Invasive Plant Specialist
P.O. Box 173120
Montana State University
Bozeman, MT 59717-3120
Phone: (406) 994-5513
Fax: (406) 994-3933
Email: [email protected]
Web: landresources.montana.edu
Noelle Orloff
Associate Extension Specialist
P.O. Box 173120
Montana State University
Bozeman, MT 59717-3120
Phone: (406) 994-6297
Fax: (406) 994-3933
Email: [email protected]
Web: diagnostics.montana.edu

Common chemical and trade names are used in this publication for clarity by the reader. Inclusion of a common chemical or trade name does not imply endorsement of that particular product or brand of herbicide. Recommendations are not meant to replace those provided in the label. Consult the label prior to any application.
Original Fall 2016 PDF (2MB)
